Recently, measles outbreaks have rebounded in many parts of the world, with the situation in the United States being particularly severe. Surveillance data show that 1,136 measles cases have been reported in the United States since 2026, and the number of cases reported in the first 8 weeks alone is six times the total annual number in previous years. As the outbreak continues to spread in Japan, Mexico and other countries, their measles elimination status has been revoked, and the global pressure of measles prevention and control has increased significantly.
Based on its independently developed core technology of TargetSeq® liquid-phase probe capture, iGeneTech has launched a measles virus liquid-phase hybridization capture kit to support prevention and control.
Background
Measles is a highly contagious acute respiratory infectious disease caused by the measles virus. Clinically, it is characterized by fever, conjunctivitis, runny nose, Koplik’s spots, generalized maculopapular rash, bran-like desquamation and brown pigmentation after rash subsidence.
The pathogenesis is as follows: the measles virus invades the body through respiratory droplets, initially replicates in respiratory epithelial cells and local lymphoid tissues, then enters the bloodstream to form the first viremia, spreads to the systemic mononuclear phagocyte system and proliferates massively, and re-enters the bloodstream to form the second viremia, resulting in extensive involvement of the skin, mucous membranes, respiratory tract, ocular conjunctiva and multiple organs throughout the body.
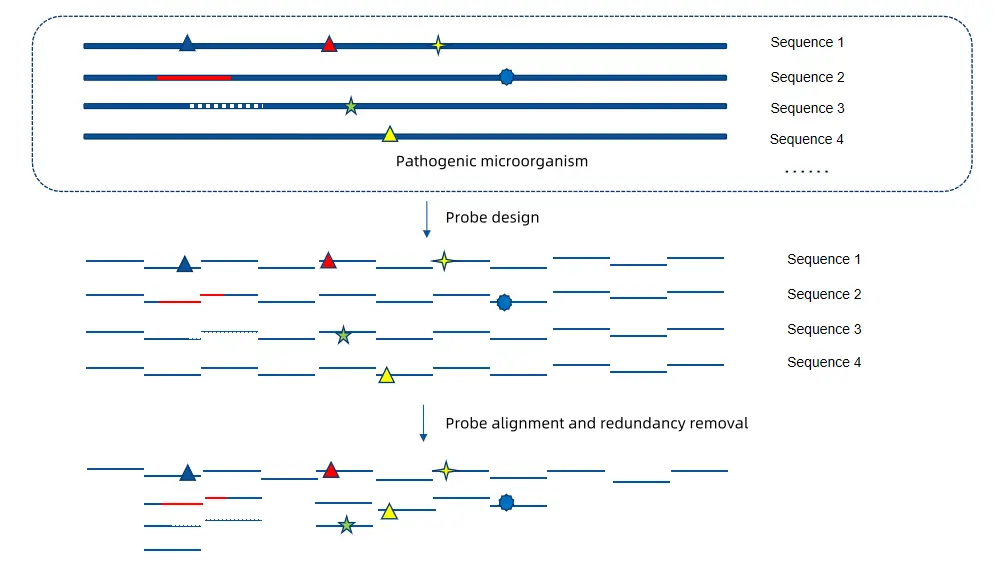
Based on its independently developed TargetSeq® liquid-phase probe capture core technology, iGeneTech has rapidly developed a measles virus liquid-phase hybridization capture kit. Targeting 1,044 full-length genomic sequences of measles virus strains collected in the NCBI public database, and adopting a high-density tiled design strategy, we finally constructed a capture probe library of 5,692 specific probes with a coverage rate of over 99.9%.
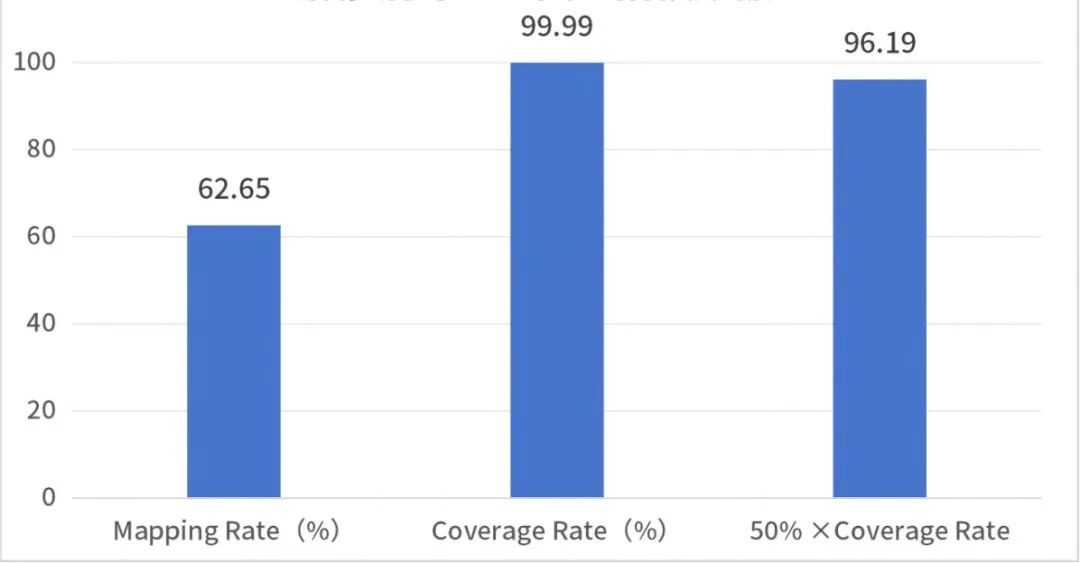
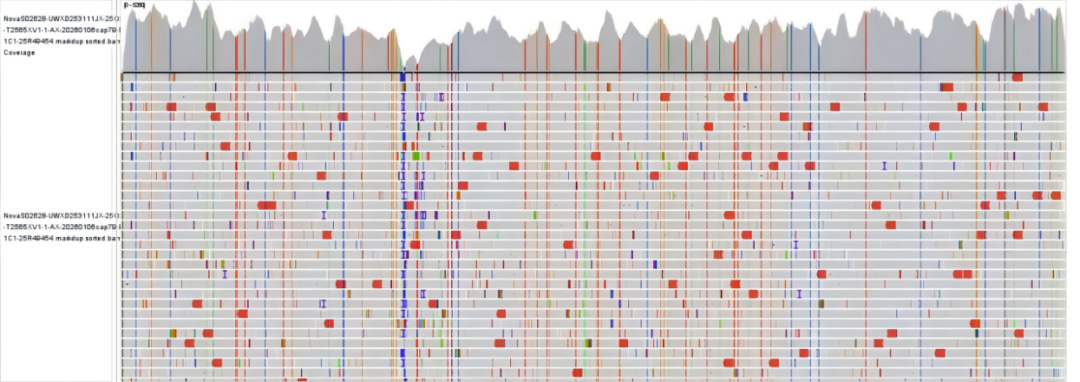
Figure 1 Measles virus whole-genome capture test data and sequencing depth plot
This technology can efficiently capture new mutation sites generated during the rapid transmission of the virus, provide high-resolution virus mutation monitoring and traceability analysis capabilities for the public health surveillance system, offer key technical support, and help achieve precise epidemic prevention and control.
Product Advantages
1. Broad sample compatibility, no cultivation required
Supports various raw sample types including human samples such as nasopharyngeal swabs, urine and serum. No pre-cultivation or host depletion steps are needed, which greatly simplifies the pre-processing workflow and significantly improves detection efficiency.
2. Broad strain coverage, complete and reliable data
Based on high-efficiency probe hybridization capture and enrichment technology, accurate and unbiased identification and detection of sample genomes can be achieved.
3. Excellent sensitivity to capture weak signals
Addressing the core challenge of low-load infections, probe optimization enables efficient binding and enrichment of target sequences. Even trace amounts of pathogen nucleic acids can be stably captured, laying a solid foundation for high-sensitivity downstream detection.
Hassle-free full-process detection: The measles virus whole-genome capture probe kit is in sufficient stock. Paired with magnetic bead-based extraction kits and RNA pathogen library construction & capture kits, it provides a seamless workflow from sample to data. It supports batch processing with automated liquid handling workstations and is compatible with multiple high-throughput sequencing platforms to meet the needs of different scenarios.
Related Product Matrix: Full-process support from extraction to detection
Product Name | Specification | Cat. No. |
Measles Morbillivirus Panel | 16/96 rxn | PH2012701/PH2012702 |
Magnetic Beads Based Pathogen DNA/RNA Co-Extraction Kit | 50 rxn | E10021 |
Magnetic Beads Based Pathogen DNA/RNA Co-Extraction Kit (Host Depletion) | 50 rxn | E20011 |
IGT® RNA Pathogen Microbial Library Prep & Capture Kit (Illumina) | 16 rxn | C11371 |
IGT® RNA Pathogen Microbial Library Prep & Capture Kit (MGI) | 16 rxn | C11441 |
IGT® Pathogen Microbial DNA & RNA Co-library Prep & Capture Kit (Illumina) | 16 rxn | C11891 |
IGT® Pathogen Microbial DNA & RNA Co-library Prep & Capture Kit (MGI) | 16 rxn | C11901 |
IGT-AS12 Automated Liquid Handling Workstation (Configuration 3) | Configuration 3 | Q91013 |

 CN
CN