Foreword
iGeneTech launches a norovirus whole-genome capture probe detection solution, supporting both manual and automated workflows, compatible with multiple sample types, and enabling efficient and accurate detection, genotyping, and source tracing.
Background
Norovirus infection is a common intestinal infectious disease, classified as other infectious diarrheal disease under Class C infectious diseases.
The virus primarily enters the human body via the oral route, targets and infects small intestinal villus epithelial cells, and replicates massively inside the cells. This leads to damage to intestinal villus structure and reduced absorption function. By disrupting intestinal water-salt metabolism, the virus causes secretory and osmotic diarrhea. It also stimulates the intestinal mucosa to release signals, activating the vomiting center through the gut-brain reflex and resulting in severe vomiting.
Norovirus infection peaks every year in winter and spring, with higher infection rates in crowded settings such as schools and kindergartens.
Infection is accompanied by mild inflammatory response, dominated by acute gastrointestinal symptoms, and is self-limiting. However, the virus has a long shedding period, making sustained transmission likely.
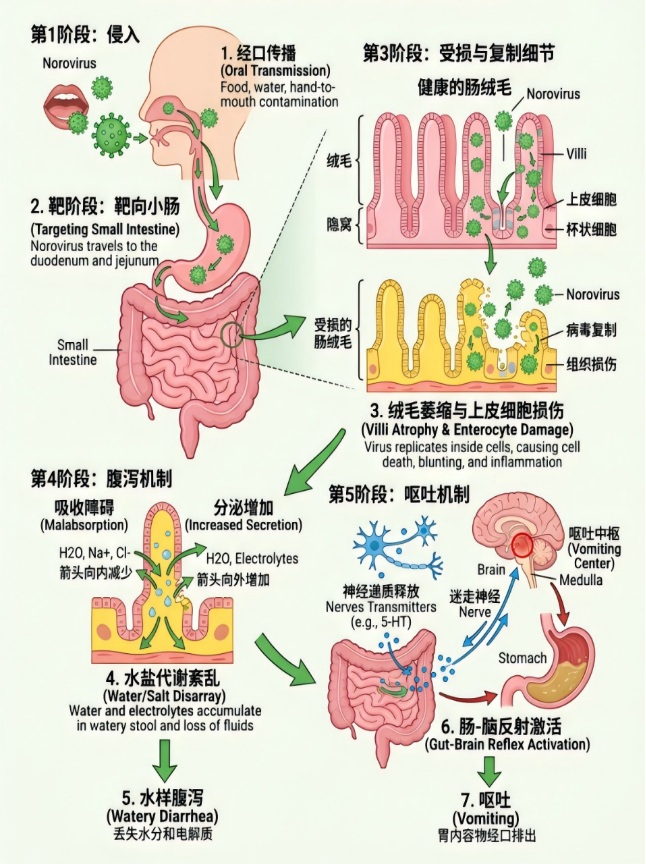
Figure 1 Schematic pathological mechanism of norovirus infection
About iGeneTech Solution
Based on its self-developed TargetSeq® liquid-phase probe capture technology, iGeneTech has developed a norovirus liquid-phase hybridization capture kit.
Using the full-length genomic sequences of Norovirus GI, GII, GIII, GIV, GV, and norovirus isolates deposited in the NCBI database over the past 20 years as target regions, the solution references 6,569 sequences and designs a total of 73,161 specific probes.
Norovirus test data
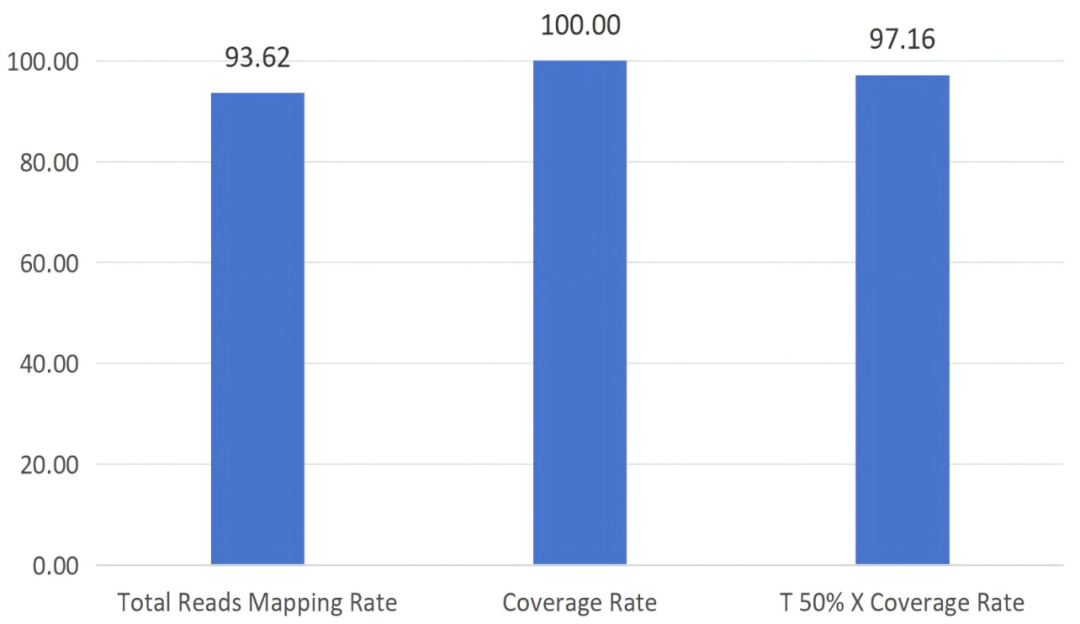
Figure 2 Whole-genome capture test data of norovirus
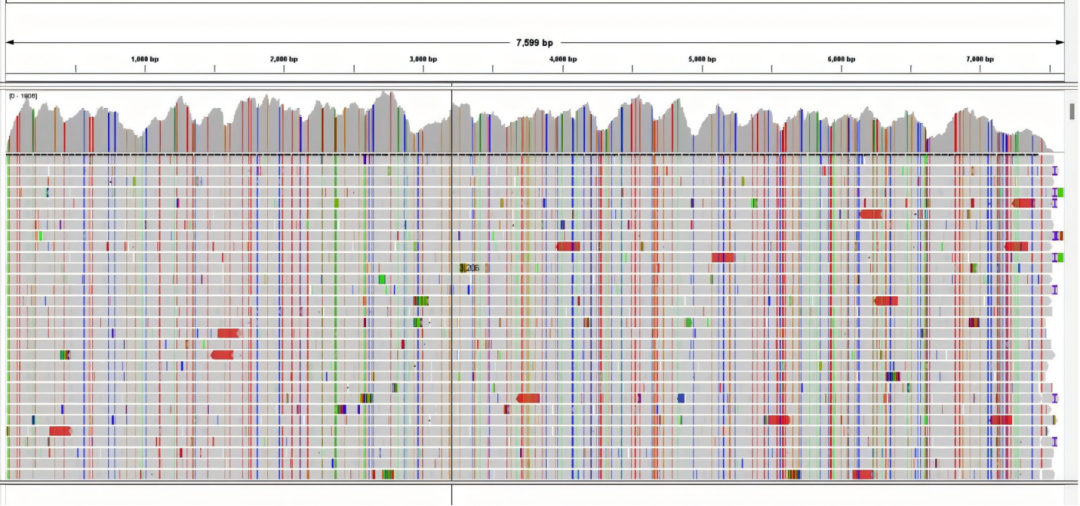
Figure 3 Norovirus sequencing depth plot
Automated Workflow
iGeneTech’s self-developed automated workstation reagent strip system enables rapid, independent processing of single samples and flexible throughput expansion via multi-unit matrix arrays, greatly reducing manual operation. Users only need to add nucleic acid (after sample extraction), capture probes, and index sequences to the reagent strip to start the fully automated process with no additional manual intervention. This fully meets laboratory demands for operational flexibility and minimal reporting turnaround time.

Figure 4 Schematic diagram of AS01 automated workstation
Based on iGeneTech’s automated pathogen whole-genome analysis pipeline, raw sequencing data undergoes quality control. Remaining reads are aligned against a pathogen reference database; reads mapped to the target pathogen are extracted and assembled de novo. The top 1 strain is selected by read count as the reference for reference-based assembly, finally generating a consensus sequence.
Product Advantages
01 Broad-spectrum, high-sensitivity, accurate and reliable
Covers the full pathogen genome. Optimized probe design stably captures trace pathogen nucleic acid. Combined with high-efficiency hybridization enrichment technology, it achieves precise genome coverage.
02 Simplified workflow, efficient and convenient
Supports multiple sample types including wastewater, human samples (feces, anal swabs, vomit), and environmental samples. No pre-culture or host depletion required. Paired with automated workflows, it greatly simplifies pre-processing and shortens detection cycles.
03 Highly integrated full process, unattended random access testing
Integrated reagent strip combines library preparation and capture. Runs independently for single samples, supporting sample-in to library-out operation. Supports multi-instrument matrix expansion with flexible throughput adaptation.
04 Physically isolated contamination control, compact deployment for stringent environments
Independent reaction units isolate cross-contamination. The compact unit occupies only 0.1 m² and can be placed directly in a biological safety cabinet, meeting high-level prevention and control requirements.
Product Matrix
Product Name | Specification (rxn) | Cat. No. |
Norovirus Panel | 16 / 96 | PH2001391 / PH2001392 |
Magnetic Beads Based Pathogen DNA / RNA Co-Extraction Kit | 50 | E10021 |
Magnetic Bead Method Pathogenic DNA/RNA Extraction Kit | 50 | E20011 |
IGT® DNA Pathogen Microbial Library Prep & Capture Kit(Illumina) | 16 | C11361 / C11431 |
IGT® RNA Pathogen Microbial Library Prep & Capture Kit(Illumina) | 16 | C11371 / C11441 |
IGT® AS12 Automated Liquid Handling Workstation (Config 3) | Config 3 | Q91013 |

 CN
CN




