Encephalitis-meningitis syndrome is an infectious syndrome caused by pathogens such as bacteria and viruses that invade the human body through various routes, cross the blood-brain barrier, and attack the central nervous system. Pathogen replication in the brain, coupled with an inflammatory storm triggered by an excessive immune response of the body, leads to meningeal edema, increased intracranial pressure and neuronal damage, resulting in symptoms such as headache and disturbed consciousness. Severe cases can cause brain herniation and even death.
Species List
Based on its independently developed TargetSeq® liquid phase hybrid capture sequencing technology, iGeneTech has developed a panel for identifying 69 common pathogenic microorganisms associated with encephalitis-meningitis syndrome, with reference to multiple strain sequences included in the NCBI database.
Among them, the probe design for 36 species of bacteria, fungi and parasites follows the principle of intraspecific conservation and interspecific specificity, while the probe design for 33 species of viruses adopts the full-length coverage strategy. A total of 218,280 probes have been designed, covering 69 common pathogens causing encephalitis and meningitis infections. This panel can be used to strengthen the surveillance of the meningitis pathogen spectrum in populations, provide important support for epidemic trend assessment, early outbreak identification and risk analysis, and meet the routine prevention and control needs of disease control departments.
Viruses (33 species)
Japanese encephalitis virus, Tick-borne encephalitis virus, West Nile virus, St. Louis encephalitis virus,Herpes simplex virus, Cytomegalovirus, Poliovirus, Varicella-zoster virus,Measles virus, Mumps virus, Rabies virus, Human herpesvirus 6,JC virus, Nipah virus, Coxiella burnetii, Human T-cell leukemia virus type 1,Powassan virus, Murray Valley encephalitis virus, La Crosse virus, Colorado tick fever virus,Western equine encephalitis virus, Eastern equine encephalitis virus, Parvovirus B19, Lymphocytic choriomeningitis virus,Influenza virus, Enterovirus, Hantavirus, Venezuelan equine encephalitis virus,Epstein-Barr virus, HIV, Hendra virus, Dabie bandavirus,Parainfluenza virus
Bacteria (19 species)
Neisseria meningitidis, Streptococcus pneumoniae, Streptococcus agalactiae, Haemophilus influenzae,Escherichia coli, Klebsiella pneumoniae, Mycobacterium tuberculosis, Staphylococcus aureus,Brucella melitensis, Treponema pallidum, Leptospira interrogans, Mycoplasma pneumoniae,Tropheryma whipplei, Bartonella, Enterococcus, Listeria monocytogenes,Group A Streptococcus, Streptococcus suis, Borrelia burgdorferi
Fungi (11 species)
Cryptococcus neoformans, Cryptococcus gattii, Aspergillus fumigatus, Aspergillus flavus,Candida albicans, Candida glabrata, Rhizopus oryzae, Absidia corymbifera,Histoplasma capsulatum, Blastomyces dermatitidis, Coccidioides immitis
Parasites (6 species)
Plasmodium falciparum, Toxoplasma gondii, Entamoeba histolytica, Taenia solium,Echinococcus granulosus, Angiostrongylus cantonensis
Experimental Workflow
1 Nucleic Acid Extraction (0.5h)
2 Library Preparation (2.5h)
3 Probe Capture (3h)
(Supplementary: Library-Probe Hybridization 0.5-1h)
4 Sequencing (3h)
5 Data Analysis
Sequencing duration is calculated based on the GeneMind FASTASeq S series sequencer with the SE100 sequencing strategy.
Product Advantages
1. Broad Sample Compatibility, No Cultivation Required
Supports a variety of raw sample types including sewage, human-derived samples (cerebrospinal fluid, nasopharyngeal swabs, blood, etc.) and environmental samples. No pre-cultivation or host depletion steps are needed, which greatly simplifies the preprocessing workflow and significantly improves detection efficiency.
2. Broad Strain Coverage, Complete and Reliable Data
Based on high-efficiency probe hybrid capture and enrichment technology, accurate and unbiased identification and detection of sample genomes can be achieved.
3. Excellent Sensitivity to Capture Weak Signals
Addressing the core challenge of low-load infections, probe optimization enables efficient binding and enrichment of target sequences. Even trace amounts of pathogen nucleic acids can be stably captured, laying a solid foundation for high-sensitivity detection in downstream processes.
4. Efficient and Fast Hybridization Process
Based on TargetSeq® liquid phase hybrid capture technology, the hybridization of libraries and probes takes only 0.5-1 hour. Combined with rapid nucleic acid extraction, library preparation and sequencing processes, the detection cycle is greatly shortened.
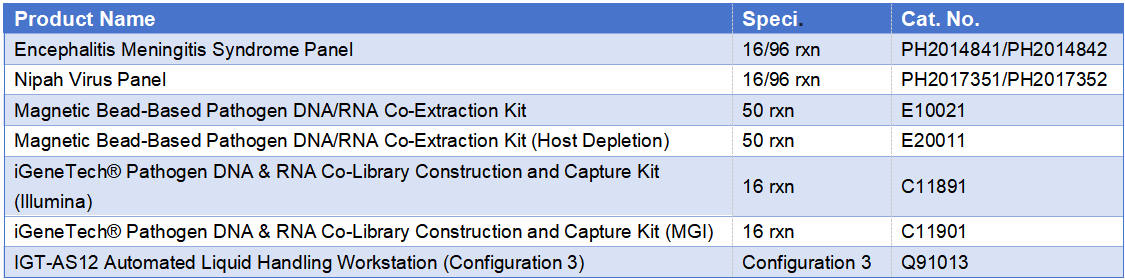
Hassle-free Full-Process Detection: Sufficient stock of the encephalitis-meningitis syndrome identification probe kit is available. Combined with magnetic bead-based extraction kits, RNA pathogen library construction and capture kits, and DNA pathogen library construction and capture kits, the workflow achieves seamless connection from sample to data. It supports batch processing with automated liquid handling workstations and is compatible with a variety of high-throughput sequencing platforms to meet the needs of different scenarios.
Related Product Matrix: Full-Process Support from Extraction to Detection

 CN
CN