Research Highlights
A team from Zhongshan Hospital, Fudan University, published a study entitled Immune repertoire sequencing for precision diagnosis in kidney transplantation in Journal of Translational Medicine.
Using TCR and BCR sequencing on peripheral blood mononuclear cells (PBMC) from patients after kidney transplantation, the study revealed characteristic immune repertoire patterns in T cell–mediated rejection (TCMR), antibody–mediated rejection (ABMR), and mixed rejection. This is the first systematic exploration of the value of immune repertoire sequencing for precise classification of renal transplant rejection.
iGeneTech is proud to have provided the immune repertoire profiling solution for this study, offering new insights into rejection monitoring and anti–rejection therapy for kidney transplant recipients.

Research Background: Biopsy Is Invasive and Has Early Blind Spots
Renal transplant rejection is the major barrier to long–term graft survival, and is mainly categorized into TCMR, ABMR, and mixed rejection. Currently, needle biopsy is the gold standard for diagnosing rejection, but it is invasive and limited in detecting early rejection.
Thus, there is an urgent clinical need for a non–invasive and precise diagnostic method.
Key Research Findings: TCR/BCR Clonal Expansion Defines Precise Classification of Renal Transplant Rejection
The research team compared immune repertoire sequencing data with biopsy pathology in post–transplant patients with undiagnosed conditions, and systematically revealed the correspondence between TCR/BCR clonal expansion and rejection types.
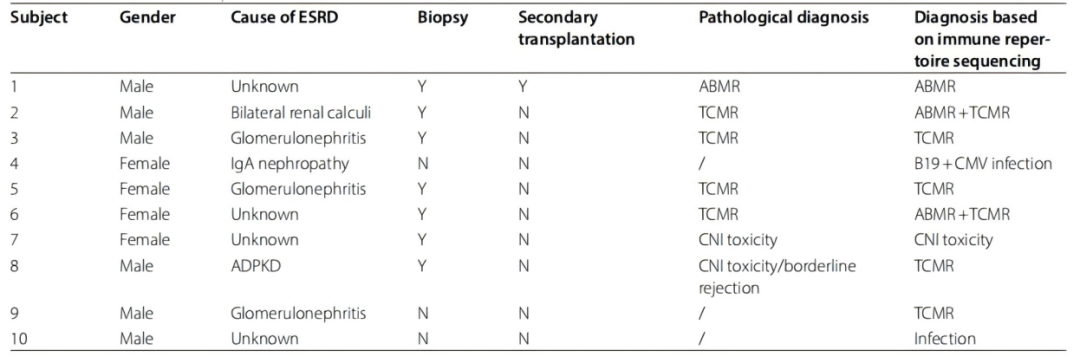
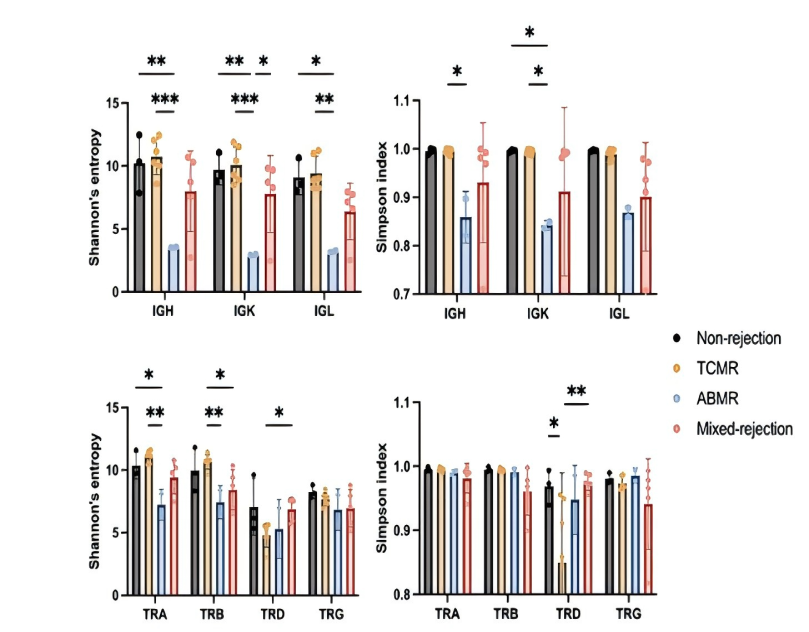
Immune Repertoire Changes Are Highly Associated with Rejection Types
By sequencing peripheral blood immune repertoires in 10 patients with graft dysfunction, researchers identified distinct TCR/BCR clonal expansion patterns across rejection types:
· TCMR patients showed marked expansion of at least one dominant TCR clone.
· ABMR patients displayed similar BCR clonal expansion patterns.
· Mixed rejection patients exhibited abnormal expansion of both TCR and BCR.
High Consistency Between Immune Repertoire Diagnosis and Biopsy Pathology
Immune repertoire sequencing achieved an overall diagnostic concordance of 80% with biopsy pathology, with dominant clonal expansion matching rejection types. Notably, sequencing corrected biopsy findings in two patients:
· Patient 2: biopsy diagnosed TCMR; sequencing indicated mixed rejection.
· Patient 8: biopsy showed borderline rejection; sequencing diagnosed TCMR.Subsequent clinical courses confirmed the sequencing diagnoses were correct.
Clinical Implications and Future Outlook: Immune Repertoire Guides Precise Rejection Classification
The expansion level and diversity of dominant TCR/BCR clones in peripheral blood are promising non–invasive warning indicators for assessing rejection and immune injury.
Dynamic tracking of dominant T/B cell clones in peripheral blood using immune repertoire sequencing may enable non–invasive, precise monitoring of post–transplant rejection, reshaping current clinical management that relies heavily on renal needle biopsy and kidney function markers.
Product Information
iGeneTech immune repertoire products are designed based on IMGT database sequences, with specific primers targeting the V and J regions of the TCR/BCR complementarity–determining region 3 (CDR3). Multiplex PCR is used to amplify the CDR3 region and evaluate changes in the immune status of samples.
Product Name | Specification (rxn) | Cat. No. |
MultipSeq® Library Prep Kit V2 | 16 / 96 | M61121 / M61122 |
IGT® UDI Primer 1–96* | 96 | C80202 |
MultipSeq® Human TCR Research Assay | 16 / 96 | M62021 / M62022 |
MultipSeq® Human BCR Research Assay | 16 / 96 | M62041 / M62042 |

 CN
CN



