
Foreword
iGeneTech has launched a detection solution for hantavirus and a multi-pathogen mixed whole-genome capture probe panel, supporting both manual and automated workflows and compatible with multiple sample types. This solution provides technical support for infectious disease prevention and control in closed spaces, enabling efficient and accurate detection, genotyping, and source tracing.
Background
On May 2, 2026, a suspected hantavirus outbreak occurred on a cruise ship in the Atlantic Ocean. Hantavirus is a zoonotic viral respiratory disease caused by viruses of the genus Orthohantavirus in the family Hantaviridae within the order Bunyavirales.
Hantavirus Pulmonary Syndrome (HPS) is characterized by headache, dizziness, chills, fever, myalgia, and gastrointestinal symptoms such as nausea, vomiting, diarrhea, and abdominal pain, followed by dyspnea and hypotension. Symptoms of HPS typically appear 2–4 weeks after initial exposure, but may occur as early as 1 week or as late as 8 weeks post-exposure.
Hantavirus primarily infects endothelial cells of small blood vessels throughout the body, causing direct endothelial damage. It also triggers a strong immune inflammatory response, forming immune complexes, activating complement, and releasing large amounts of inflammatory factors. This leads to a sharp increase in vascular permeability, resulting in plasma extravasation, edema, and hypotensive shock. The virus can impair platelet function, disrupt coagulation mechanisms, induce DIC (Disseminated Intravascular Coagulation), and cause systemic bleeding tendencies.
Based on its independently developed TargetSeq® liquid-phase probe capture core technology, iGeneTech has developed a hantavirus liquid hybridization capture kit. Using 1,378 full-length hantavirus genome sequences from the NCBI public database over the past 20 years as target regions, the kit adopts a high-density tiled design to construct a capture probe library containing 48,191 specific probes.
This technology efficiently captures newly emerging mutation sites generated during rapid viral transmission, providing high-resolution monitoring of viral variation and source-tracing capabilities for public health surveillance systems. It delivers critical technical support for precise epidemic prevention and control.
Hantavirus Test Data
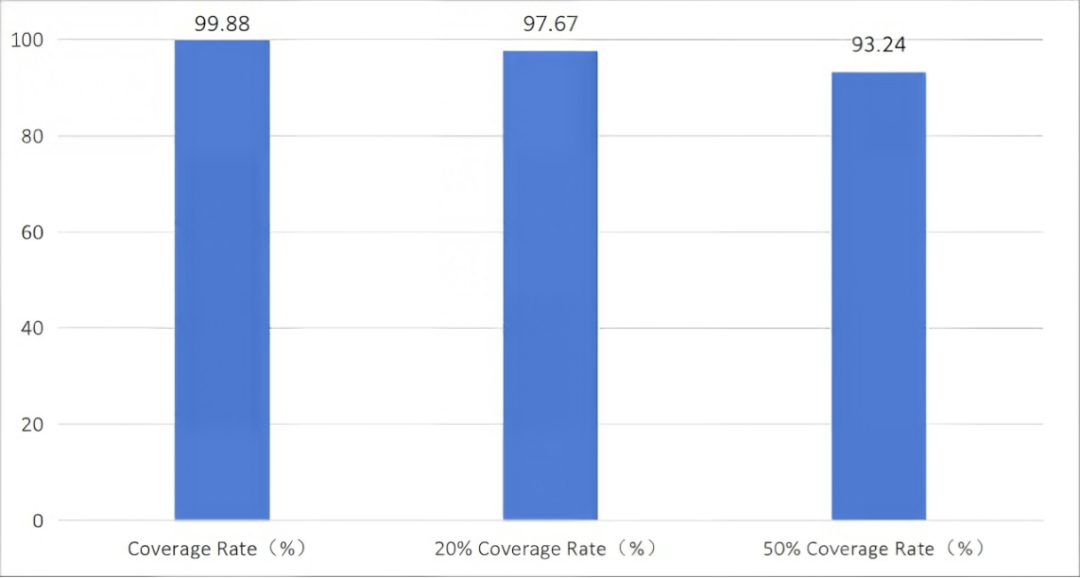
Figure 1 Whole-genome capture data of Hantavirus
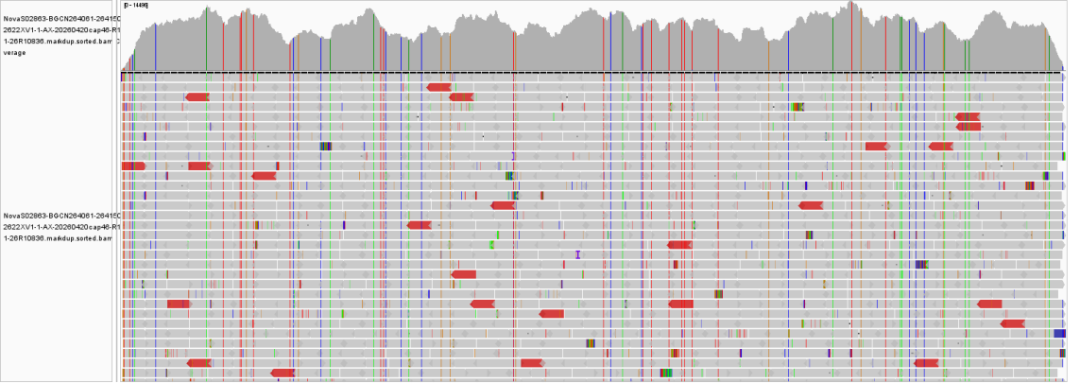
Figure 2 Sequencing depth plot of Hantavirus
In highly crowded, relatively closed public settings such as cruise ships, there is a risk of cross-transmission of multiple pathogens. To address this demand, iGeneTech has built a pathogen monitoring system suitable for such environments, supporting combined detection of samples from ship blackwater, mixed domestic sewage, sediments, and key environmental areas.
Meanwhile, the iGeneTech team has independently developed a multi-pathogen mixed whole-genome capture probe panel, covering common pathogenic microorganisms including influenza virus, SARS-CoV-2, Mycoplasma pneumoniae, norovirus, enterovirus, monkeypox virus, and hantavirus. A total of 372,938 specific probes have been designed to enable simultaneous enrichment and accurate detection of multiple pathogens, supporting infectious disease control in closed spaces.
Automated Workflow
iGeneTech’s independently developed automated workstation reagent strip system enables fast, independent processing of single samples and flexible throughput expansion via multi-unit arrays, greatly reducing manual operation. Users only need to add nucleic acid (extracted from samples), capture probes, and index sequences to the reagent strips to start the fully automated process with no additional manual intervention, fully meeting laboratory needs for operational flexibility and rapid reporting.

Figure 3 Schematic diagram of AS01 automated workstation
Based on iGeneTech’s automated whole-genome analysis workflow for pathogenic microorganisms, raw sequencing data undergoes quality control. Remaining reads are aligned against a pathogen reference database; reads mapped to target pathogens are extracted and assembled de novo. The top 1 strain is selected by read count as the reference for reference-based assembly, finally generating a consensus sequence.
Product Advantages
01 Broad-spectrum, High-sensitivity, Accurate and Reliable
Covers the full pathogen genome; optimized probe design stably captures trace pathogenic nucleic acids. Combined with high-efficiency hybridization enrichment, it achieves precise genome coverage.
02 Simplified Workflow, Efficient and Convenient
Supports multiple sample types including sewage, human samples (feces, anal swabs, vomit), and environmental samples. No pre-culture or host depletion is required. Paired with automated workflows, it greatly simplifies pre-processing and shortens detection cycles.
03 Fully Integrated, Unattended, Walk-up Testing
Integrated reagent strips combine library preparation and capture. Single-sample independent operation enables “sample-in, library-out” processing. Supports multi-machine array expansion with flexible throughput.
04 Physically Isolated to Prevent Contamination, Compact for Harsh Environments
Independent reaction units eliminate cross-contamination. The compact unit occupies only 0.1 m² and can be placed directly in a biological safety cabinet, meeting high-level biosafety requirements.
Product Matrix
| Product Name | Specification (rxn) | Catalog No. |
| Hantaviridae Panel | 16 / 96 | PH2007571 / PH2007572 |
| Magnetic Beads Based Pathogen DNA / RNA Co-Extraction Kit | 50 | E10021 |
| Magnetic Bead Method Pathogenic DNA/RNA Extraction Kit | 50 | E20011 |
| IGT® DNA Pathogen Microbial Library Prep & Capture Kit(Illumina) | 16 | C11361 / C11431 |
| IGT® RNA Pathogen Microbial Library Prep & Capture Kit(Illumina) | 16 | C11371 / C11441 |
| IGT-AS12 Automated Liquid Handling Workstation (Config 3) | Config 3 | Q91013 |

 CN
CN




